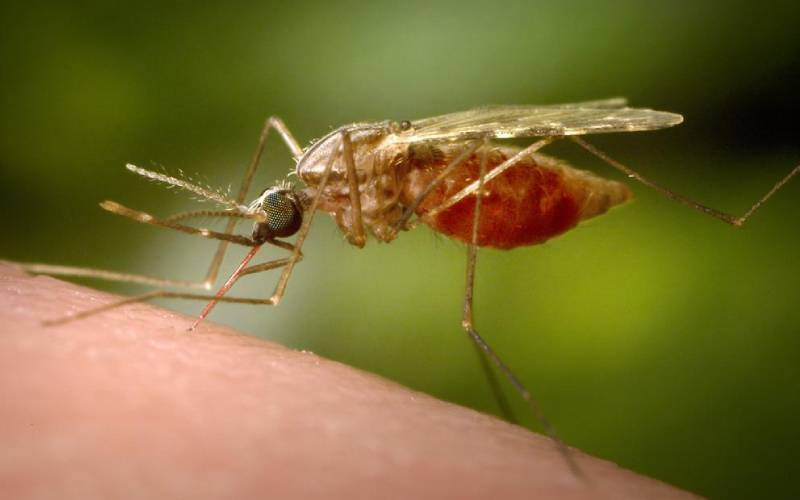
Research in Africa found a one-time dose of an experimental drug protected adults against malaria for at least six months, the latest approach in the fight against the mosquito-borne disease.
Malaria killed more than 620,000 people in 2020 and sickened 241 million, mainly children under 5 in Africa. The World Health Organization is rolling out the first authorized malaria vaccine for children, but it is about 30 per cent effective and requires four doses.
The new study tested a very different approach - giving people a big dose of lab-made malaria-fighting antibodies instead of depending on the immune system to make enough of those same infection-blockers after vaccination.
"The available vaccine doesn't protect enough people," said Dr Kassoum Kayentao of the University of Sciences, Techniques and Technologies in Bamako, Mali, who helped lead the study in the villages of Kalifabougou and Torodo.
In those villages during malaria season, other research has shown, people are bitten by infected mosquitoes on average twice a day.
The experimental antibody, developed by researchers at the U.S. National Institutes of Health, was given by IV - difficult to deliver on a large scale. But the encouraging findings bode well for an easier-to-administer shot version from the same scientists that's in early testing in infants, children and adults.
The U.S. government research was published Monday in the New England Journal of Medicine and presented at a medical meeting in Seattle.
- How one father is leading Kenya's malaria fight
- How raw milk and red meat drive brucellosis transmission in humans
- Researchers find malaria's weak point, offer hope for world children
- Advocates demand to know the truth about GMO mosquitoes in Nairobi
Keep Reading
The antibody works by breaking the life cycle of the parasite, which is spread through mosquito bites. It targets immature parasites before they enter the liver where they can mature and multiply. It was developed from an antibody taken from a volunteer who received a malaria vaccine.
The research involved 330 adults in Mali who got either one of two different antibody doses or a dummy infusion. All were tested for malaria infection every two weeks for 24 weeks. Anyone who got sick was treated.
Infections were detected by blood test in 20 people who got the higher dose, 39 people who got the lower dose and 86 who got the placebo.
The higher dose was 88 per cent effective, compared to the placebo. The lower dose was 75 per cent effective.
Protection might last during the several months of a malaria season. The idea is to someday use it alongside other malaria prevention methods such as malaria pills, mosquito nets and vaccines. Cost is uncertain, but one estimate suggests lab-made antibodies could be given for as little as $5 per child per malaria season.
Lab-made antibodies are used to treat cancer, autoimmune diseases and COVID-19, said Dr. Johanna Daily of Albert Einstein College of Medicine in New York, who was not involved in the study.
"The good news is now we have another, immune-based therapy to try to control malaria," Daily said.
 The Standard Group Plc is a multi-media organization with investments in media
platforms spanning newspaper print
operations, television, radio broadcasting, digital and online services. The
Standard Group is recognized as a
leading multi-media house in Kenya with a key influence in matters of national
and international interest.
The Standard Group Plc is a multi-media organization with investments in media
platforms spanning newspaper print
operations, television, radio broadcasting, digital and online services. The
Standard Group is recognized as a
leading multi-media house in Kenya with a key influence in matters of national
and international interest.











