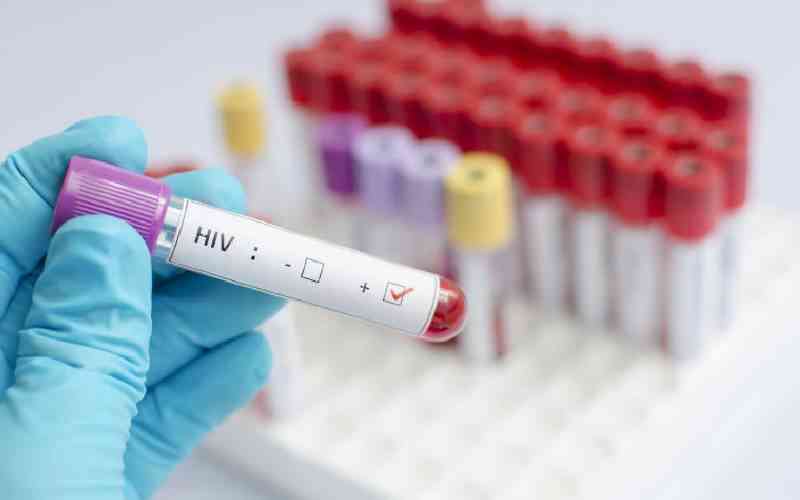
It is good news for millions of Kenyans living with HIV/AIDS as Kenya will start buying new highly-effective antiretroviral drugs at much lower prizes following an international price deal.
The deal negotiated by Kenya and South Africa together with a number of international organisations will see the new HIV drugs cost not more than Sh650 per month per person or about Sh7700 per year (US$75).
About 1.5 million Kenyans have HIV and the government provides free drugs to more than a million of them with the help of donors. Now it will cost far less to purchase latest HIV drugs, meaning more people can be reached for treatment.
Kenyans with HIV will access a single-pill HIV treatment regimen containing Dolutegravir (DTG), a new drug that has been in use in high-income countries to combat HIV but was unaffordable to low and medium-income countries.
Dolutegravir is a highly effective antiretroviral, which is well tolerated by patients and has fewer side effects and resistance compared to HIV drugs currently being used in Kenya.
“In the antiretroviral therapy guidelines launched in July 2016, the Ministry of Health made provisions for use of newer antiretroviral medicines such as DTG,” said Dr. Cleopa Mailu, Cabinet Secretary of Health.
He added: “Research has shown that DTG offers better tolerability, fewer adverse drug reactions, fewer drug interactions, and higher genetic barrier to resistance. It is with this in mind that, in July this year, Kenya approved its inclusion in the National antiretroviral treatment Programme,”
The agreement was reached by the governments of Kenya and South Africa together with international organisations including the Clinton Health Access Initiative (CHAI), the Joint United Nations Programme on HIV/AIDS (UNAIDS), the Bill & Melinda Gates Foundation (BMGF).
Others are the United Kingdom’s Department for International Development (DFID), the United States’ President’s Emergency Plan for AIDS Relief (PEPFAR), the U.S. Agency for International Development (USAID), and the Global Fund to Fight AIDS, Tuberculosis and Malaria together with Mylan Laboratories Limited and Aurobindo Pharma,
The new drug is widely used in high-income countries since 2014 and is recommended by the World Health Organization (WHO) as an alternative first-line HIV regimen.
The drug is also expected to reduce the need for more expensive second- and third-line regimens.
The drug will be available as a pill to be taken once daily combining DTG together with other anti-HIV drugs tenofovir and lamivudine.
It was developed by Mylan and Aurobindo under licensing agreements from ViiV Healthcare, the original developer of the drug.
New orders for the drugs are expected to begin early next year. The price agreement applies to public sector purchases in 92 countries.
In July this year, the Ministry of Health got 148,000 packs of the generic drug dolutegravir (DTG) worth Sh60 million to supply 27,000 Kenyans who have developed resistance to Efavirenz, a component of the first-line HIV drug in the country. The drugs were donated by UNAITAID, an international NGO.
The drug has been available in local private hospitals at between Sh5000 and Sh6,000 for a 30-day supply pack.
“This agreement will improve the quality of life for millions of people living with HIV,” said UNAIDS Executive Director Michel Sidibé. “To achieve treatment targets, newer, affordable and effective treatment options must be made available without any delay.”
 The Standard Group Plc is a multi-media organization with investments in media
platforms spanning newspaper print
operations, television, radio broadcasting, digital and online services. The
Standard Group is recognized as a
leading multi-media house in Kenya with a key influence in matters of national
and international interest.
The Standard Group Plc is a multi-media organization with investments in media
platforms spanning newspaper print
operations, television, radio broadcasting, digital and online services. The
Standard Group is recognized as a
leading multi-media house in Kenya with a key influence in matters of national
and international interest.