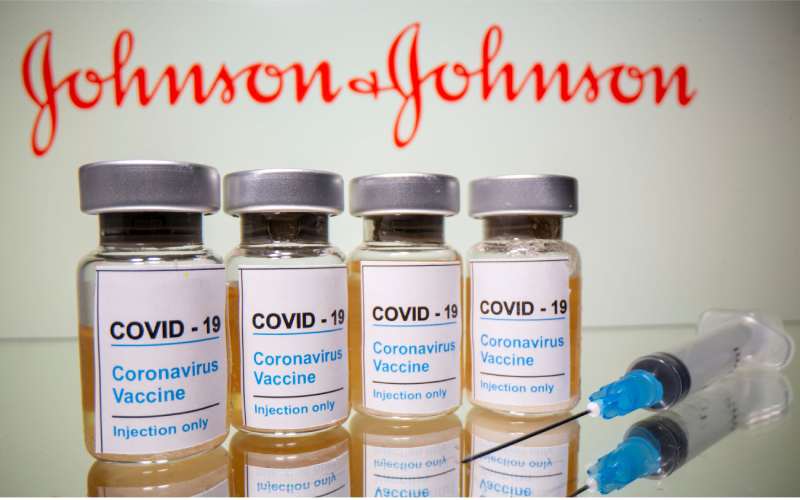
The United States federal health agencies on Tuesday recommended pausing the use of Johnson & Johnson’s single-dose COVID-19 vaccine. This is after six recipients developed a rare disorder involving blood clots.
The U.S. Centre for Disease Control and Prevention will hold an advisory meeting on Wednesday to review the cases.
All the six recipients were women between the ages of 18 and 48. One woman died and a second in Nebraska has been hospitalized in critical condition, the New York Times reported.
The CDC and the U.S. Food and Drug Administration said in a joint statement that the adverse events appear to be extremely rare right now.
As of April 12, more than 6.8 million doses of the Johnson & Johnson vaccine have been administered in the United States.
The company’s shares were down 3% before the opening bell.
The move from the U.S. regulators comes less than a week after Europe’s drugs regulator said it was reviewing rare blood clots in four people in the United States who received the shot.
 The Standard Group Plc is a multi-media organization with investments in media
platforms spanning newspaper print
operations, television, radio broadcasting, digital and online services. The
Standard Group is recognized as a
leading multi-media house in Kenya with a key influence in matters of national
and international interest.
The Standard Group Plc is a multi-media organization with investments in media
platforms spanning newspaper print
operations, television, radio broadcasting, digital and online services. The
Standard Group is recognized as a
leading multi-media house in Kenya with a key influence in matters of national
and international interest.