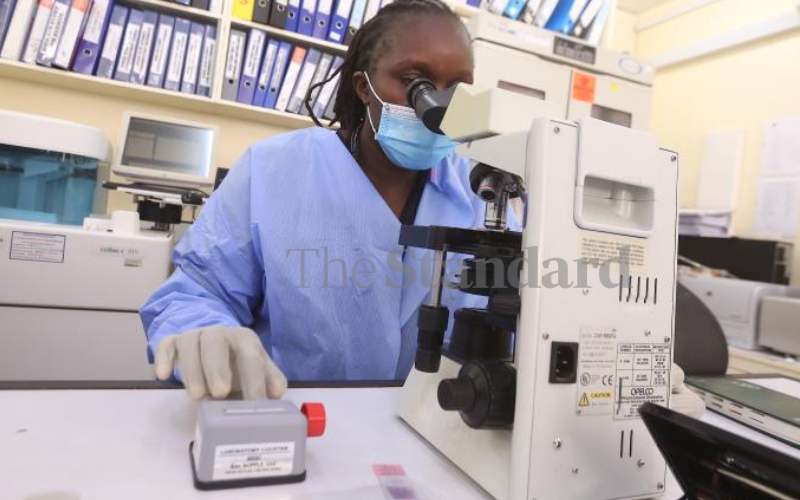
Lab technologists at Kombewa Clinical Research Centre in Seme, Kisumu County. [Collins Oduor, Standard]
Kenya suffers a huge burden of diseases with a low number of clinical researches.
There is only one certified facility in Nairobi with a capacity to facilitate phases 1 and 2 of clinical trials and though it needs urgent expansion, it relies on grants, instead of regular government support.
According to Novartis Group Head, Corporate Affairs and Global Health, Dr Lutz Hegemann, the situation has many elements needing consideration in terms of availability of clinical investigators and willing participants for the trials “to ensure the medicines are appropriate and developed in the region.”
Dr Bernhards Ogutu, a clinical research scientist at the Kenya Medical Research Institute (Kemri) notes that the underrepresentation of Africa in clinical trials could only mean the findings can hardly be termed as universal.
Ogutu explained that to have a successful clinical trial, there needs to be the right infrastructure including a clinic area and a laboratory with a supportive referral system and an enabling regulatory framework for a seamless workflow of clinical trials.?
He cited few clinical research coordinators among other cadres needed for clinical trials as another setback and “for a long time we have had external investigators coming, making monitoring clinical trials and studies very expensive.”
Novartis in partnership with Kemri, for instance, is assessing the efficacy and safety of two doses of Crizanlizumab therapy in adolescents and adults living with Sickle Cell Disease.
Dr Hegemann argues that there is a need to screen newborns, diagnose them and provide first-line treatment with a possible solution of advanced therapies and Novartis is willing to partner with sickle cell foundations, governments, diagnostic companies to complement efforts of creating this ecosystem.
Such partnerships will help wean the continent of donations to a model that makes medicines, available, affordable, and of high quality and later strengthens the health systems.
Dr Ogutu also reckons that relying on donor funding shifts focus from local problems to global problems and “sadly for a long time African governments have been more focused on curatives activities and research has not been biomedically centred.”
Dr Ogutu adds that most trials are carried out on imported drugs, yet “drugs we get must be optimised to the right dosage because different groups of people handle drugs differently” and that African countries do not consider themselves as part of the solutions but dedicated consumers.
For wrestling down malaria, Novartis Pharmaceuticals has partnered with KEMRI Walter-Reed Kombewa research site, to develop a drug combination for treating severe malaria in children.
The novel non-artemisinin combination - ganaplacide/lumefantrine was carried in two sites of Kombewa and Siaya in children under 12 and Dr Hegemann says this once-daily dose combination “has the potential not only to clear malaria infection, including artemisinin-resistant strains but also to block the transmission of the malaria parasite.”
Dr Hegemann observed the trials showed that the combination of ganaplacide/lumefantrine solid dispersion formulation was generally well tolerated in children.
He explained that children below 5kg body weight are disproportionately affected by malaria: “Even with our existing drug combination therapy we have found a reformulation of two active ingredients to make it more suitable for newborn babies whose bodies operate differently from adults or more grown children.”
He stated medicines used to treat patients with sickle cell and malaria are not enough and there is a need to look into the entire ecosystem and “we do this by listening and talking to the physicians while looking at the societal needs while complementing the technical bit that comes with the medicine and other interventions that are needed which require partnerships.” He adds: “We would love to see malaria eliminated in the coming years, and sickle cell disease in children, where they can live a productive life. Advances in science can help address the matter.”
 The Standard Group Plc is a multi-media organization with investments in media
platforms spanning newspaper print
operations, television, radio broadcasting, digital and online services. The
Standard Group is recognized as a
leading multi-media house in Kenya with a key influence in matters of national
and international interest.
The Standard Group Plc is a multi-media organization with investments in media
platforms spanning newspaper print
operations, television, radio broadcasting, digital and online services. The
Standard Group is recognized as a
leading multi-media house in Kenya with a key influence in matters of national
and international interest.