WHO agrees on protocols to start trials on Covid-19 herbal remedies
Health & Science
By Graham Kajilwa| 5yrs ago | 3 min read
.
Popular this week
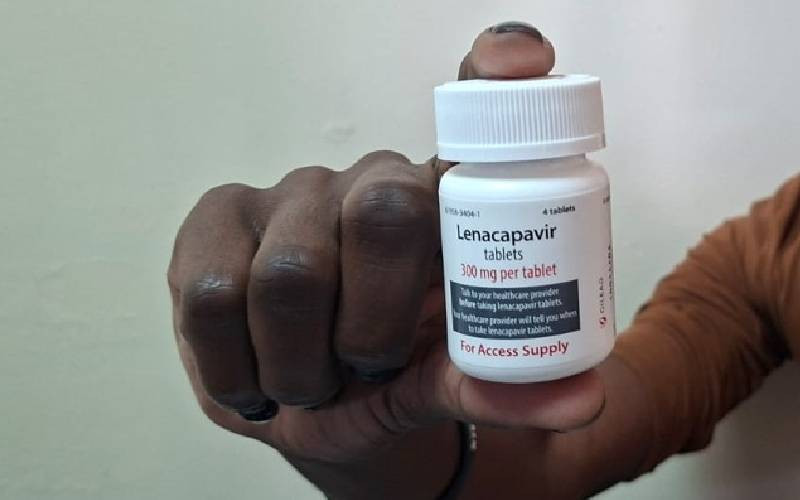
- Kenya rolls out six- month HIV injection, easing daily pill pain
- Bare reality: How acute condom shortage fuels surge in HIV, STIs
- Breach of Privacy: PSK condemns secret recording of Gachagua at pharmacy
- Experts from 15 nations complete Kenya-led IAEA course
- Eating udongo during pregnancy may kill your child’s learning ability
- Punda amechoka
- Kenyan police officer to lead continental association
- Ruto calls for education stakeholders collaboration to overcome CBE challenges
- New drug to boost women’s sex drive approved
- 25 million kids missed routine vaccinations because of Covid-19
Previous article
.
Similar Articles
.
Latest Articles
Health & Science
2026-02-27 08:50:00
Health & Science
By Mark Oloo
2026-02-26 20:30:44
Health & Science
By Mate Tongola
2026-02-26 17:47:26
Health & Science
By Ryan Kerubo
2026-02-26 17:04:00
.
 The Standard Group Plc is a multi-media organization with investments in media
platforms spanning newspaper print
operations, television, radio broadcasting, digital and online services. The
Standard Group is recognized as a
leading multi-media house in Kenya with a key influence in matters of national
and international interest.
The Standard Group Plc is a multi-media organization with investments in media
platforms spanning newspaper print
operations, television, radio broadcasting, digital and online services. The
Standard Group is recognized as a
leading multi-media house in Kenya with a key influence in matters of national
and international interest.