The poisons board has clarified that a hypertension drug once linked to cancer is safe.
Hydrochlorothiazide, an over the counter antihypertensive drug has been cleared by the Pharmacy and Poisons Board (PPB) as safe and with no known such cancer causing effects.
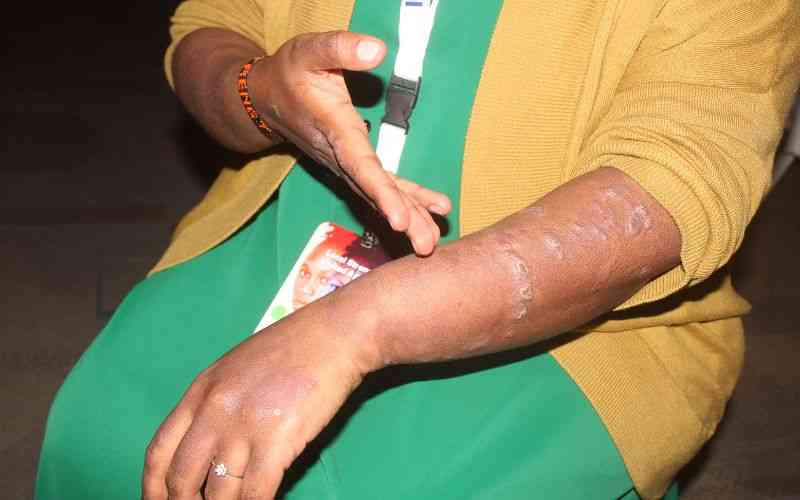
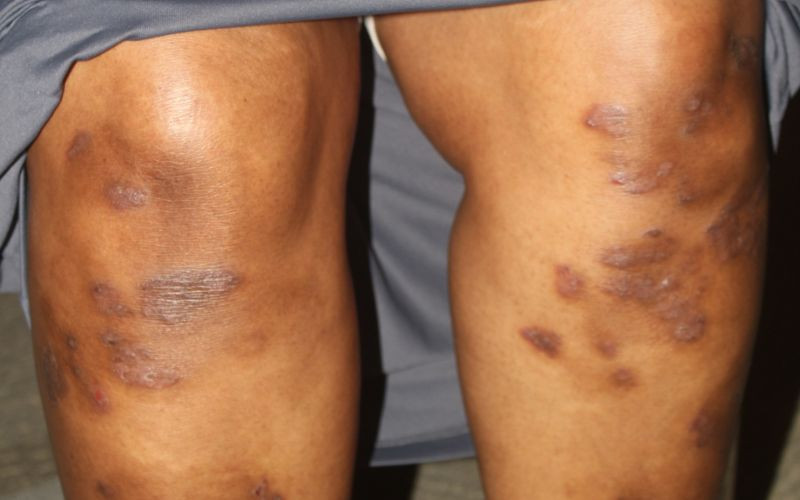
An earlier study had linked users of the drug to increase susceptibility to skin cancer.
However, PPB in its update on Thursday said the study was done predominantly on white population who are already susceptible to skin cancer because of their skin colour.
"Therefore, there is no strong evidence to warrant the stoppage of use of HCTZ in our local setting," said the board's Chief Executive Officer Fred Siyoi.
Siyoi said nevertheless, health care providers are advised to encourage the patients not to stop their medication but to look out for any suspicious lesions on their skin and report to the nearest health facility or health care provider.
"In addition, they (health care providers) need to take proper history of patients so that those who may be genetically predisposed to skin cancer because of family history are given alternative medicines," he said.
Siyoi said the caution by PPB is a routine post authorization safety update based on epidemiological studies that reported increase in the risk of the Adverse Drug Reactions (ADRs).
The board explained that the data of the ADR does not warrant the recall or withdrawal of the products rather calls for restrictions in prescribing and closer monitoring of patients using this group of medicines.
"As medicines continue to be used by large populations, adverse effects that were not picked out during clinical trials emerge. Any significant safety updates on medicines such as those of the above mentioned have to be updated on the patient information leaflet/ or medicines insert," he said.
All this information should be reported to PPB.
The board in July 2018, had also issued an alert on a recall instituted on some products containing valsartan-also found in some hypertension drugs.
The recall, said PPB, was instituted after reports of contamination of specific valsartan active pharmaceutical ingredient manufactured in different countries and used to manufacture valsartan containing products
Valsartan, is an active pharmaceutical ingredient (API) used either alone or in combination with other active pharmaceutical ingredients for the treatment of hypertension.
"The recall affected only those products and batches that contained the affected valsartan active ingredient that had been contaminated," he said.
Valsartan containing products that were manufactured using the Active Pharmaceutical Ingredients sourced at Zhejiang Huahai Pharmaceuticals, based in Linhai, China.
"There are other valsartan products in the market not affected by the recall hence your healthcare provider will guide you appropriately," said the board's boss.
He added: "PPB reminds the public that these medicines should only be obtained on prescription from a qualified medical practitioner."
 The Standard Group Plc is a multi-media organization with investments in media
platforms spanning newspaper print
operations, television, radio broadcasting, digital and online services. The
Standard Group is recognized as a
leading multi-media house in Kenya with a key influence in matters of national
and international interest.
The Standard Group Plc is a multi-media organization with investments in media
platforms spanning newspaper print
operations, television, radio broadcasting, digital and online services. The
Standard Group is recognized as a
leading multi-media house in Kenya with a key influence in matters of national
and international interest.