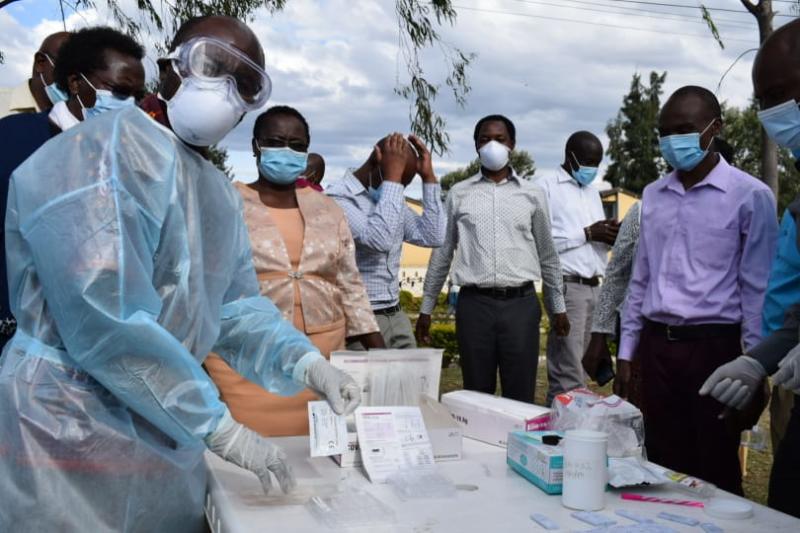
Medical laboratory technologists in Nakuru trained on rapid antigen tests. [ Mercy Kahenda, Standard]
The introduction of antigen testing in the country is among measures aimed at boosting the fight against coronavirus.
The technology has first been adapted in Nakuru County, in collaboration with Clinton Health Access Initiative (CHAI).
The introduction of the technology comes at the time the Ministry of Health has documented 1,167,409 cumulative tests.
Yesterday, 130 people tested positive for the disease, out of 4, 918 samples.
“From the cases, 115 are Kenyans while 15 are foreigners,” read a Covid-19 statement by the ministry.
One patient succumbed to the virus, bringing total deaths to 1,751 since the pandemic was reported in the country in March last year.
Country Director for CHAI Mr Gerald Macharia said 2,000 rapid antigen testing kits will be donated to Nakuru County to kick start the testing.
“CHAI will also support the training of 60 Medical Lab Technologists on the Covid-19 Rapid Antigen testing,” he said.
Rapid antigen testing is a diagnostic test that directly detects the presence or absence of a specific protein (antigen) that causes Covid-19.
The technology does not require a laboratory for the processing of results, which are processed within 15 minutes.
Also, with the technology, individuals who are at a greater risk of contracting the virus can easily be identified.
Nakuru Chief Officer of Public Health Samuel King’ori said the technology will boost the fight against the spread of the virus.
“Delay in release of tested samples has been a major hitch in contact tracing and surveillance. It has been taking about three or five days to receive results,” said the official.
The county has trained 30 medical laboratory technologists from Level Four hospitals, and the Rift Valley Provincial General Hospital, to kick off the testing.
At least 50 people have been tested with the technology, ahead of the main launch next week.
“With trained human resource, we shall reach out to more population in hotspot areas, to help flatten the Covid-19 curve,” added King’ori.
Nakuru Governor Lee Kinyanjui is on record pleading with the Ministry of Health to consider certifying a Covid-19 laboratory.
The county has been collecting samples and transporting them to different laboratories across the country.
Among laboratories that have been serving the county include International Livestock Research Institute (ILRI), National Influenza, and the Kenya Medical Research Institute (KEMRI) in Nairobi.
A report on Covid-19 country preparedness and response by Kenya Medical Practitioners and Dentists Council (KMPDC) in October last year recommended the Health ministry to facilitate counties seeking accreditation and certification for Covid-19 laboratory testing capabilities.
The council also recommended strengthening mechanisms for external laboratory referral services from institutions such as Moi Teaching and Referral Hospital, Kenya Medical Research Institute (Kemri), and National Public Health Laboratories (NPHL) for Covid-19 sample testing.
The country has a total of 41 accredited laboratories for testing Covid-19, out of which, 12 are private ones.
In April, the ministry promised that each of the 47 counties would have an equipped laboratory with the capacity to test for Covid-19.
According to the ministry, testing would have been made possible after the Pharmacy and Poisons Board (PPB), the country’s regulator for medicines and health technologies reduced the period for certifying test kits and other equipment used to test the disease.
Health Cabinet Secretary Mutahi Kagwe said PPB had already authorised the use of four test kits.
The kits are COBAS SAR –Covid-2 Test kit by Roche Diagnostics, Xpert Xpress SARS CoV-2 by Cepheid, Biofire Covid-19 Test by Biofire Defence LLC, and Abbott by Abbott Molecular Inc.
Moi Teaching and Referral Hospital was the latest centre to be activated to test for Covid-19, which serves about 22 counties.
The CS said the board was implementing stringent, expedited accreditation mechanism, which has shrunk the process of certifying test kits from three months to 48 hours.
Dr Ahmed Kalebi, CEO and Chief Consultant Pathologist at Lancet Group Laboratories, East Africa told The Standard that testing should take between 24 and 48 hours.
 The Standard Group Plc is a multi-media organization with investments in media
platforms spanning newspaper print
operations, television, radio broadcasting, digital and online services. The
Standard Group is recognized as a
leading multi-media house in Kenya with a key influence in matters of national
and international interest.
The Standard Group Plc is a multi-media organization with investments in media
platforms spanning newspaper print
operations, television, radio broadcasting, digital and online services. The
Standard Group is recognized as a
leading multi-media house in Kenya with a key influence in matters of national
and international interest.