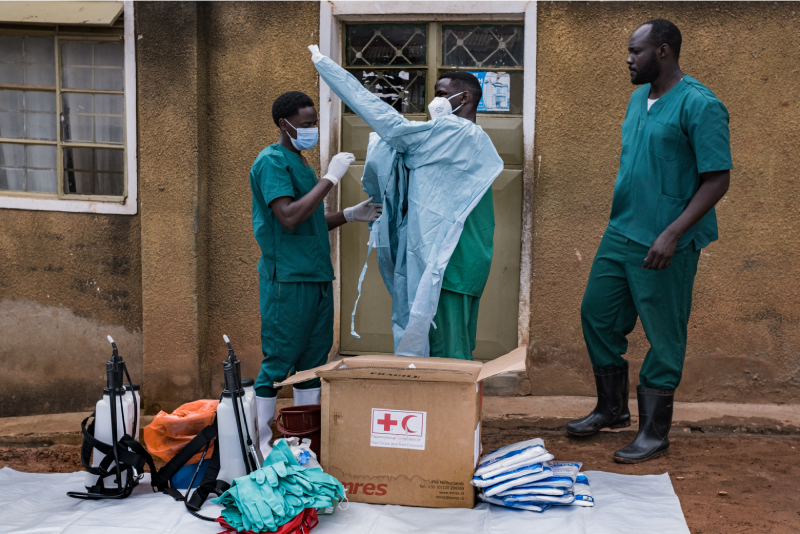
Recruitment of 400 volunteers for Covid-19 vaccine trial is set to begin in Kilifi County.
The process is expected to kick off on October 1.
The Kenya Medical Research Institute (Kemri) that is conducting the exercise, says a few housekeeping issues are still being worked on before the process begins.
“We have received all the required national approvals and now we are pursuing consent from the study sites and then we are good to go,” says Kemri Director-General Yeri Kombe.
This will make Kenya the second country in the continent after South Africa to engage in Covid-19 vaccine trial.
The study will be carried out at the Kilifi County Hospital, but other sites in Mombasa County may be included as the work progresses.
Social requirements
The volunteers will have to meet strict medical and social requirements for the two-year study. For example, women must not be breastfeeding or pregnant and not planning to conceive within the study period.
They will be required to produce evidence that they are on an effective contraceptive or be in a strictly monogamous relationship where the male is either sterilised or using an effective birth control method.
“In such case, the male partner should have been sterilised at least six months prior to the female subject’s entry into the study and the relationship is monogamous,” says the study protocol.
If the male partner is using a condom as a contraceptive, which has about two per cent failure rate, the woman should also be on a spermicide or a diaphragm.
People with a drinking problem are also locked out of the study. “Suspected or known current alcohol abuse; that is taking more than two bottles of 500mls beer a day or more than two large glasses of 12 per cent wine per day are not eligible,” says the protocol.
Seven clinic visits
The volunteers, who will be women and men aged over 18, must be healthy, with no chronic diseases or suppressed immunity, HIV negative and with no bleeding disorders or cancer.
The study will be open only to frontline workers who include health workers, truckers and others as defined by the Ministry of Health.
The ministry defines immigration, custom and security officers as frontline workers. Other frontline workers may include community health volunteers, support staff in hospitals and some cadres in the travel and hospitality industry.
Once recruited, 200 participants will be injected with the experimental Covid-19 vaccine while a similar number will be put on a rabies vaccine, which acts as the control.
Out of these, 40 will be enrolled in phase Ib trial, and the remainder, 360, in phase II of the study.
The participants will be followed closely, especially for the first 28 days after the vaccine. “Each participant will have seven clinic visits in the course of the study for medical assessment,” says the protocol. Consequently, participants are expected to live within the study area for the trial period.
Kemri says volunteers will be reimbursed costs of participation such as travel costs.
Compensation is fixed
“The compensation is fixed so that volunteers are not out of pocket as a result of their participation with the rate dependent on various factors, including transportation, the time required to participate and other expenses incurred by the volunteer,” Kemri told The Standard in an email.
Media reports in the US and UK where the same vaccine is being tested indicate participants may be receiving $1,000 (about Sh100,000) and £650 (about Sh90,000) respectively for the whole study duration.
But local participants, Kemri says, will also benefit by knowing their general health status, including whether they are infected with SARS-CoV-2.
“If global data demonstrate the safety and efficacy of this trial vaccine, the control group that received a rabies vaccine will be offered the ChAdOx1 nCOV-19 vaccine.”
The government is represented in the study by the Chief Administrative Secretary for Health Rashid Aman, who is also a researcher.
Initially, the study was to start in May but has since been going through the approval process by which time trials in the UK, Brazil and the US have moved to Phase III.
Then the infection rate of Covid-19 was very high at the Coast but has since plummeted, which could cause a challenge for the trials.
 The Standard Group Plc is a multi-media organization with investments in media
platforms spanning newspaper print
operations, television, radio broadcasting, digital and online services. The
Standard Group is recognized as a
leading multi-media house in Kenya with a key influence in matters of national
and international interest.
The Standard Group Plc is a multi-media organization with investments in media
platforms spanning newspaper print
operations, television, radio broadcasting, digital and online services. The
Standard Group is recognized as a
leading multi-media house in Kenya with a key influence in matters of national
and international interest.